  
They are commonly used by chemists to decide the strength of chemical reactants in laboratories, and by pharmacists to understand the dosage of drugs. 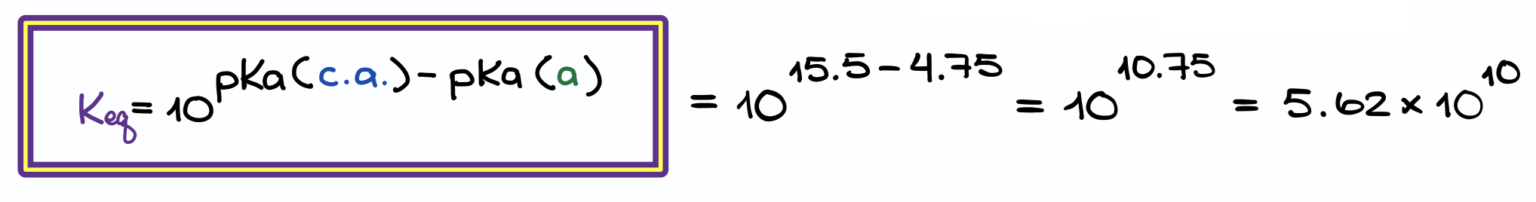
While pH is a popular value used to indicate the number of protons that are produced by a substance, there are other quantities, like K a, K b, pK a, and pK b, used to explain the strength of these substances. This is how the Brønsted-Lowry Theory describes the concept of an acid and a base. Based on how eagerly these substances donate or accept such protons, they are categorized as ‘strong’ or ‘weak’. On the other hand, a base is a substance which readily accepts such free protons. By this principle, an acid is a substance which readily donates its hydrogen ions (called protons) when dissolved in water. The chemical nature of any substance depends on the charged particles that it produces, called ‘ions’. Moreover, acids and bases always neutralize each other. Despite the general notion that only strong acids can burn the skin, strong bases also have the same effect. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2022
Categories |
 RSS Feed
RSS Feed
